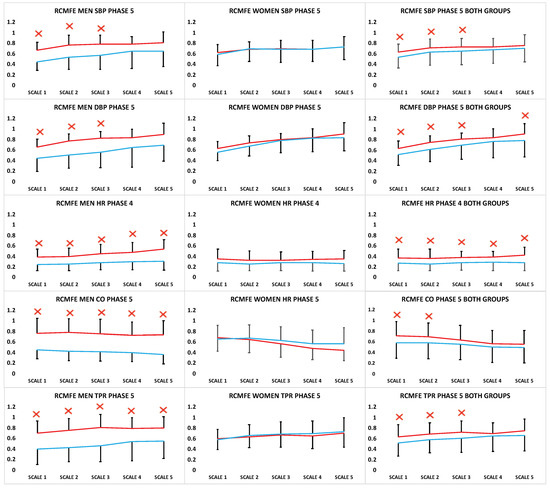
In the case of gas particles, Entropy is generally higher when compared to solid ones. To apply the second law we consider the total. The lost work ( in Equation ( 6.4 )) is always greater than zero, so the only way to decrease the entropy of a system is through heat transfer.
CHANGE IN ENTROPY FORMULA FREE
For example, in case of solid where particles are not free to move frequently, the Entropy is less as compared to gas particles that can be disarranged in a matter of minutes. The content of Equation ( 6.4) is that the entropy of a system can be altered in two ways: (i) through heat exchange and (ii) through irreversibilities.

Entropy is basically a thermodynamics function that is needed to calculate the randomness of a produce or system.
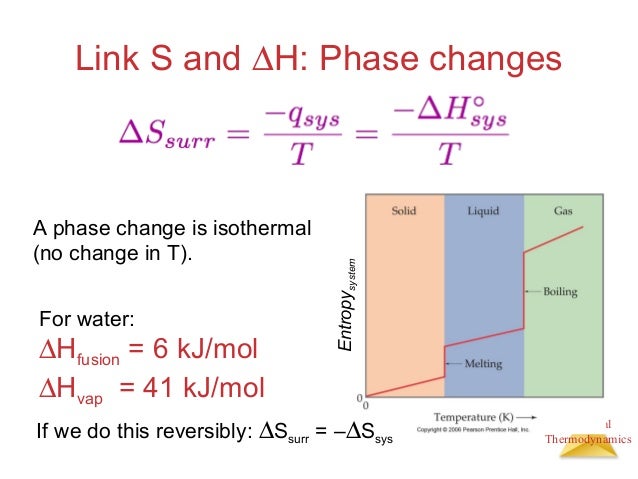
This was second lay of thermodynamics where the concept of Entropy came into existence. I hope you must have a better idea of Entropy here with this example. So, from an ordered stage, perfume reached to disordered stage by spreading throughout the room. dU TdS + PdV (change in energy temperature change in entropy + pressure change in volume) This equation describes the combined effects of the. Another good example to state the definition of Entropy is Spraying perfume at one corner of the room, so what will happen next? The perfume will not stay in one corner only but its fragrance can be felt everywhere. n 1 because you have one mole of gas and R is a constant (8.3145 J/molK). V2/V1 would be 1/3 because you compress the ideal gas to one third of its original volume. This is the formula for change in entropy when temperature is constant. For dQ of irreversible the equation should be changed into the clausius inequality form which is dS> dQ (irreversible)/T. The equation of dSdQ/T is not an accurate equation, the actual equation should be dSdQ (reversible)/T. The higher the entropy, the more ways the system is disordered. I think you would use the formula deltaS nRln (V2/V1). Irreversible free expansion of a gas in adiabatic condition is not isentropic. Let us see how it works actually.Įntropy is defined as the total number of ways how a system can be arranged. Obviously, there are multiple ways to arrange the bag of ball and entropy concept is somewhat similar in Chemistry. Repeat the process until the balls are not ended in the bag. Now draw one more ball from the bag and try to find out the answer of same question. Take an example that you have a bag of balls and if you draw one ball from the bag then how many possible ways to arrange the balls together on the table.
